Last week, on the coldest, rainy and blusterously windy day, we had a half day off, lit the stove in the kitchen after lunch, and stayed inside and cooked.Janine made a ginger parkin cake in her very special Swiss expanding cake tin. This was a gift from Barbara, a potter in Switzerland when we were working there some years ago. It had been handed down in her family for some years. Barbara had cooked a cake in it and brought it along to the wood firing workshop that we were doing. I was so taken by its ingenious simplicity, I studied it and even did a drawing of it with the details and dimentions, so that I could make a copy of it for us on our return. I have learnt some very basic skill in sheet metal working. Barbara saw that I was taken by it, with me coming back to it several times and examining it and photographing it.At the conclusion of the workshop it was presented to me gift wrapped. I was really touched. I am still really impressed with this cake tin every time it comes out and Janine bakes in it. Most of all I still recall Barbara, Catherine, Stefan, Eric and the others, up in the Swiss Alps and the great time that we all had together. It brings back strong memories and feelings of good times. It’s a loaf shaped cake tin, but it has a madeleine effect.

This two piece cake tin concertinas inside itself, so it is always the perfect size for almost any size recipe.
While Janine was making her cake, I roasted hazelnuts under the grill, just enought to bring out a little colour and that amazing smell and flavour.We had spent the few nights prior in front of the idiot box shelling the nuts. I melted 2 blocks of cheap, no-frills, German, organic, supermarket 70% dark chocolate in a double boiler, made from a bowl suspended in a source pan of boiling water, I mixed the nuts into the melted chocolate, then poured the mass out into a square cake tin to cool. It is so amazingly delicious, tasty and fragrant, but not sweet, just nutty, melt in your mouth smooth, but with that special freshly roasted hazelnit crunch. Wow! We may not have any money, but we live a rich life. Imagine trying to buy this at DJ’s food hall?





It goes very nicely with coffee.

In the afternoon we made marmalade. A lovely, relaxing and rewarding way to spend a miserable day in the warm kitchen.

The next day, back in the pottery, I retrieved the anvil that had been through the fire and then sat out in the yard for 21 months waiting for some TLC.It should have been rescued much earlier, but I’m working to my limit and I don’t want to overdo it. I can’t do everything. So now is the time to retrieve the anvil. It is the worse for the weather even though I had it tarped, but fire does strange things to iron, and it rusts so much faster than if it had just be put outside. I cleaned a lot of flakey rust off the surface and then treated it with rust converter to stabbilise it. I remember when I bought it back in the 70’s, at an auction for $110. I was so much younger and I could lift it into the VW beetle back then to bring it home.I must have been mad! It’s somewhere around 100 kilos, if not more. I can’t lift it now!I had to use the little crane on my truck to pick it up, and then build a tripod and use a chain block to lift it onto its new block. It’s a thing of beauty and a joy forever! This anvil has an English maker’s stamp on it. I imagine, sometime in the 1800’s?



I cleaned all the loose scale off the surface and swept it up to use as an iron pigment in the future. I read somewhere that Hamada had said that blacksmith’s iron scale made the best iron pigment. I’ll give this a go. I’ll have to calcine it, crush it and grind it very finely first.


I have decided that the best way to use the small ball mill roller is to use it to make pigments. I have done a few experiments with it this week and found that as it overheated and shut down with the 4 litre jar that it came with. I tried my 1 litre jar and also added a small desk top fan to cool the motor. This was the combination that worked well enough, so that it can run for a few hours without over heating and shutting down. I made up a batch of a cobalt pigment that I developed over 40 years ago, just after I left Art School. It has a softer more ‘natural’ look, more like ‘gosu’, with an iron break where it is thicker. We used this recipe for 20 years until I found our local natural cobalt bearing stone.



Recipe;
Red basaltic soil 66%. – Red top soil is high in iron and silica and fuses readily.
Potash felspar 33% – The felspar helps to melt it into the surface of the glaze.
Cobalt oxide 1% – cobalt oxide is such a very very strong colorant, you only need a very small amount.
Cobalt is far too strong to use straight as an oxide. It is also rather grainy and spotty. It needs to be ‘watered down’ with something else to soften it out a bit and dilute it. I have read some pretty exotic recipies that use zinc and tin and manganese, nearly always iron and some with bone ash.
Michael Cardew recomended a mix of;
Cobalt carbonate 20%
White Tin Oxide 20%
Black manganese oxide 10%
Plastic red clay 15%
China clay 20%
red iron oxide 5%
I tried this recipe when I was a student at Art School, but didn’t like it. A bit too much like fountain pen ink.
My local red clay and cobalt recipe is simplier and it’s mine. It also has that lovely blue/black cobalt and iron break between thick and thin.
I found a deposit of natural cobalt south of here 15 years ago. It was a mix of silica, iron, manganese and only just a miniscule amount of cobalt, but it showed a pale blue when disolved onto a glaze as a brush stroke.
I have always had an interest in fossiking for local materials, processing them and intergrating them into my work. As you can see below, I tested 90 different samples of precipitates from chalybeate springs in the area of my research around here, both active and ancient and dried out and solid stone-like deposits. Numbers 84, 86 and 88. showed a blue colour. Interestingly, the top row were fired in reduction atmosphere and were definitely blue with the cobalt showing clearly.The bottom row were fired in oxidation and the cobalt blue is diluted by the manganese showing a paler grey, purple/mauve blue.Sample number 90 appears to contain mainly manganese and iron with very little cobalt present, being a pale brownish, claret/mauve colour.
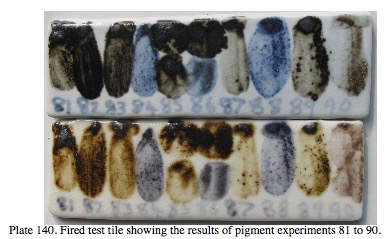
My samples averaged;
Silica 55%
Iron Oxide 1%
Manganese Oxide 16%
Cobalt oxide 1%
This equates to something like the ancient Chinese and/or Japanese ‘Gosu’ or ‘Smalt’ natural cobalt bearing pigment stones.
If nothing else, I learnt that I must always test everything in both oxidation and reduction to get a true undersanding of what is going on.
Tragically, I lost nearly all my aged porcelain stone bodies, pigments, ores and minerals in the fire, and now many of the places that I used to fossick are now sterilised by housing developments. It’s going to be a different life now, with reduced access to the minerals that I used to use. We’ll develop a more restrained and refined palette.
So I’m back to milling up a red soil and cobalt mixture to mimic the natural pigment that we used to use.

To make a glaze for my porcelain tests, I have collected and spread out to dry, some of my porcelain clay turnings in the window. I will calcine these and use them as a glaze material too. When blended with Moss Vale limestone, they will melt into a very pale, limpid porcelain glaze. – I hope!
So much to do and so little time.

You must be logged in to post a comment.